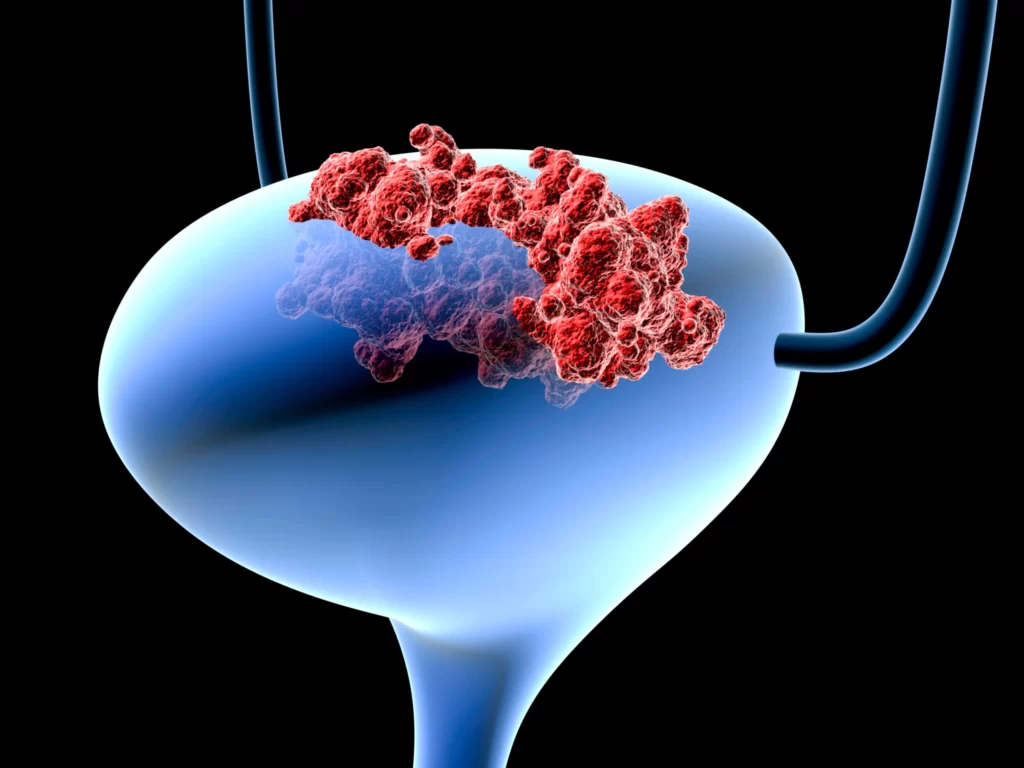
Adstiladrin is a therapy for patients with an aggressive form of the disease whose only other option is to have their bladder removed.
The treatment, which is administered into the patient’s bladder once every three months, causes the body to produce a protein that fights cancer.
The FDA stated that Adstiladrin’s safety and effectiveness were evaluated in a multi-center clinical study involving 157 patients.
Ferring stated that it is too early to comment on Adstiladrin pricing, but the company intends to make the drug widely available to patients.
According to a 2021 report from the Institute for Clinical and Economic Review, the drug provides good value for patients when priced between $158,600 and $262,000.
According to Ferring, adstiladrin will be commercially available in the United States in the second half of 2023.
Bladder cancer is one of the most common cancers in the United States, with non-muscle invasive bladder cancer (NMIBC) accounting for approximately 75% of all cases.
About Adstiladrin
Adstiladrin is a gene therapy developed as a treatment for adult patients with BCG-unresponsive NMIBC. It is a non-replicating adenovirus vector-based gene therapy containing the gene interferon alfa-2b, administered by catheter into the bladder once every three months. The vector enters the cells of the bladder wall, releasing the active gene to do its work.
The cells’ internal gene/DNA machinery “picks up” the gene and translates its DNA sequence, causing the cells to secrete large amounts of interferon alfa-2b protein, a naturally occurring protein that the body uses to fight cancer.
This novel gene therapy approach converts the patient’s own bladder wall cells into interferon micro factories, boosting the body’s natural anti-cancer defences.
To read our blog on “Biotech company is using tiny worms in a test for pancreatic cancer,” click here